Optical Coherence Tomography: New Evidence Lends Support for MS Diagnosis
By measuring retinal thinning, optical coherence tomography (OCT) provides an inexpensive and noninvasive means of assessing CNS pathology in people with MS. Two new studies show that OCT findings correlate with spinal cord and cortical MRI measures.
Over the past 30 years, magnetic resonance imaging (MRI) has emerged as a primary tool for diagnosing multiple sclerosis (MS). More recently, a new kid on the block, optical coherence tomography (OCT), has been gaining recognition as a cheaper and less invasive means of assessing pathology in the central nervous system (CNS) of MS patients.
In two presentations at the American Academy of Neurology (AAN) annual meeting in Philadelphia, researchers from Peter Calabresi, M.D.’s group at Johns Hopkins University advanced the argument that OCT generates high-resolution images that reflect global disease processes in the CNS, and that OCT measures correlate significantly with MRI measures of both spinal cord and cortical lesions, as well as with clinical disability.
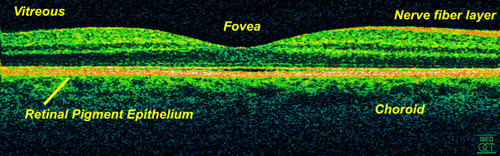
Retinal pathology correlates with spinal cord lesions
According to Jiwon Oh, M.D., a neurologist and clinical research fellow at Johns Hopkins, the optic nerve and spinal cord are both commonly involved in MS, leading to optic neuritis and sensory and motor symptoms, respectively. “From an anatomic perspective, both regions are spatially distinct, unique compartments of the CNS that have very strong structure-function relationships, and as a result, they are ideal substrates to study in order to better understand how MS disease processes cause disability in patients,” she said in her platform presentation.
Oh and colleagues enrolled 102 MS patients and 11 healthy controls in their study. Each subject underwent cervical spinal cord diffusion-tensor imaging and magnetization-transfer imaging on a 3-tesla MR scanner. Postacquisition image processing yielded a number of sensitive measures, including fractional anisotropy, a measure sensitive to axonal integrity; perpendicular diffusivity, a measure sensitive to microstructural tissue integrity; and magnetization transfer ratio, a measure of demyelination.
They also measured the spinal cord cross-sectional area, which reflects spinal cord tissue atrophy. Clinical assessments included the usual measures of global disability (the Extended Disability Status Scale, or EDSS; and the MS functional composite) as well as measures more relevant to spinal cord involvement such as hip flexion strength and vibration sensation threshold. Retinal imaging with OCT enabled measuring the thickness of multiple axonal and neuronal layers of the retina, including the retinal nerve fiber layer (RNFL), ganglion cell–inner plexiform layer (GCIP), inner nuclear layer, and outer nuclear photoreceptor layer. RNFL and GCIP thinning are greater in MS patients as a result of optic nerve pathology, but recent studies have suggested that these measures reflect global MS pathology as well.
Using multivariable linear regression adjusted for age, sex, prior optic neuritis, and brain atrophy, the researchers demonstrated “robust and consistent relationships” between many spinal cord and retinal measures in MS subjects but not in healthy controls. Surprisingly, Oh said, both the OCT retinal measures and the spinal cord measures correlated with clinical measures specific to the visual system, such as visual acuity at high and low contrast. Likewise, while vibration sensation threshold was expected to show a relationship only with spinal cord measures, the retinal measures also showed a relationship.
“These results seem to suggest that the spinal cord and retina independently reflect global pathological processes relevant to disability in MS, which measures of brain atrophy do not adequately capture,” Oh concluded. “This highlights the importance of combining measures from unique compartments of the nervous stems to facilitate a more thorough examination of the complex regional and global disease processes that contribute to clinical disability in MS.”
Cortical lesions
Daniel Harrison, M.D., also working with Calabresi, conducted a similar study, this time comparing the relationship between cortical gray matter lesions and retinal pathology in MS. Cortical gray matter lesions are found in nearly all MS patients, typically involving areas of cortical demyelination and/or neuronal loss, Harrison said in his platform presentation. “We wanted to determine whether or not cortical lesions by MRI and retinal pathology by OCT contribute independent biomarkers of the pathology that lead to disability in MS.”
Harrison and colleagues analyzed data from 12 subjects with MS and seven controls who had undergone ultrahigh-field 7-tesla (7T) MRI scans, OCT scans, structural MR measures of atrophy, and disability assessment using the EDSS. Two different 7T MR protocols enabled assessment of three subtypes of cortical lesions: leukocortical lesions transversing from white into gray matter, intracortical lesions in the gray matter, and subpial lesions, larger lesions that transverse large areas of the cortex and that are thought to arise through a different mechanism than other cortical lesion subtypes.
Although the study was small, the results showed that after adjusting for age, gender, and optic neuritis history, the cortical lesion burden correlated poorly with peripapillary RNFL thinning, suggesting that cortical lesions are not the result of axonal pathology. However, cortical lesion burden, particularly in the subpial region of the cortex, did correlate with neuronal retinal measures indicating possible neuronal degeneration.
Next, Harrison wanted to test whether these measures could be used to predict disability. He used a nested regression model with likelihood ratio testing, which allowed him to test the correlation with disability when individual measures are added together. Starting with a “base model” comprised of cortical lesion count or volume, age, gender, and history of optic neuritis, Harrison showed that the addition of neuronal retinal measures predicted disability better than cortical lesion burden alone.
An expanding role for OCT?
If validated, these studies both suggest that retinal layer thickness determined by OCT provides clinically useful information beyond what can be determined using both conventional and nonconventional MRI. “There’s really no other system like vision in the nervous system where you can so elegantly correlate structure with function,” said neurologist Laura Balcer, M.D., of New York University Langone Medical Center in an interview with MSDF. Balcer was not involved in the OCT studies presented at the AAN meeting. While OCT measures are not completely specific to MS, she said, the visual pathway can mirror what is happening globally.
As the MS diagnostic criteria continue to evolve, they may incorporate OCT measures. “That’s why these correlations are important, so that in the future we can be confident regarding the relationship of OCT to MRI, and perhaps be able to use OCT as an additional tool more upfront to help in the diagnosis,” Balcer said.
“I think we will see more and more the use of OCT in practice, and it’s through our experience in practice as well as through the trials that we learn what the changes mean for our patients,” she added. Already, OCT measures have been incorporated into clinical trials, particularly of repair agents used to treat patients with acute optic neuritis.
Key open questions
- Can retinal measures from OCT be used as outcome measures in clinical trials of disease-modifying therapies in MS?
- How are clinical changes in symptomatology and disease progression reflected in OCT measures?
Disclosures and sources of funding
Dr. Oh’s research is supported by a 2012 Decker Family endMS Transitional Career Development Award. Dr. Oh has received personal compensation for activities with EMD Serono, Genzyme Inc., Biogen Idec, and Novartis as a consultant and speaker. Some of her co-authors disclosed financial compensation from Abbott, Bayer, Biogen Idec, Diagnosoft, Genzyme, MedicalLogix, Novartis, Teva Neurosciences, and Vertex.
Dr. Harrison’s study was supported by Merck Serono. He has received personal compensation for activities with Questcor Pharmaceuticals.
Dr. Balcer has received personal compensation for activities with Biogen Idec, Questcor, Novartis, and Vaccinex.



Comments
The basis for OCT interpretation depends on computer algorithms that 'dissect' retinal layers individually and can perhaps discern between normal vs abnormal in a specific layer with great accuracy. The biggest concern for such definitive statements about retinal pathology is that alternative algorithms even if available, are not generally used to validate the analyses. Hence, what is applicable under a set of 'rules' of a particular algorithm must be validated using alternative algorithms that should come to the same conclusion and then these data sets become powerful. I am not sure that I have seen even one article that validates data analyses using different algorithms. However, this is not to suggest that the analysis is flawed - what I am stating is that they can become even more powerful if another lab or researcher in another setting using a different algorithm validates the findings keeping in mind that most OCT findings are not routinely validated by histopathology findings.
JR Avasarala